The State of Gene Therapy and ASO Development: What Families Need to Know About the Growing Field
If your child has a rare genetic disease and you are trying to understand what treatments might be available, one of the first questions you probably have is: how real is this? Are gene therapies and ASOs actually reaching patients, or is this still experimental? How many have been approved? How many are in development? And what are the chances that something could help your child?
This article answers those questions with data. It is a landscape review of the gene therapy, ASO, and gene editing field as of early 2026, written for families, not investors. The numbers are sourced from the FDA, the American Society of Gene and Cell Therapy (ASGCT), and published peer-reviewed literature.
The short version: this field is real, it is growing, and more therapies are reaching patients every year. But there are also real challenges, especially in manufacturing, that families should understand.
How Many Therapies Have Been Approved?
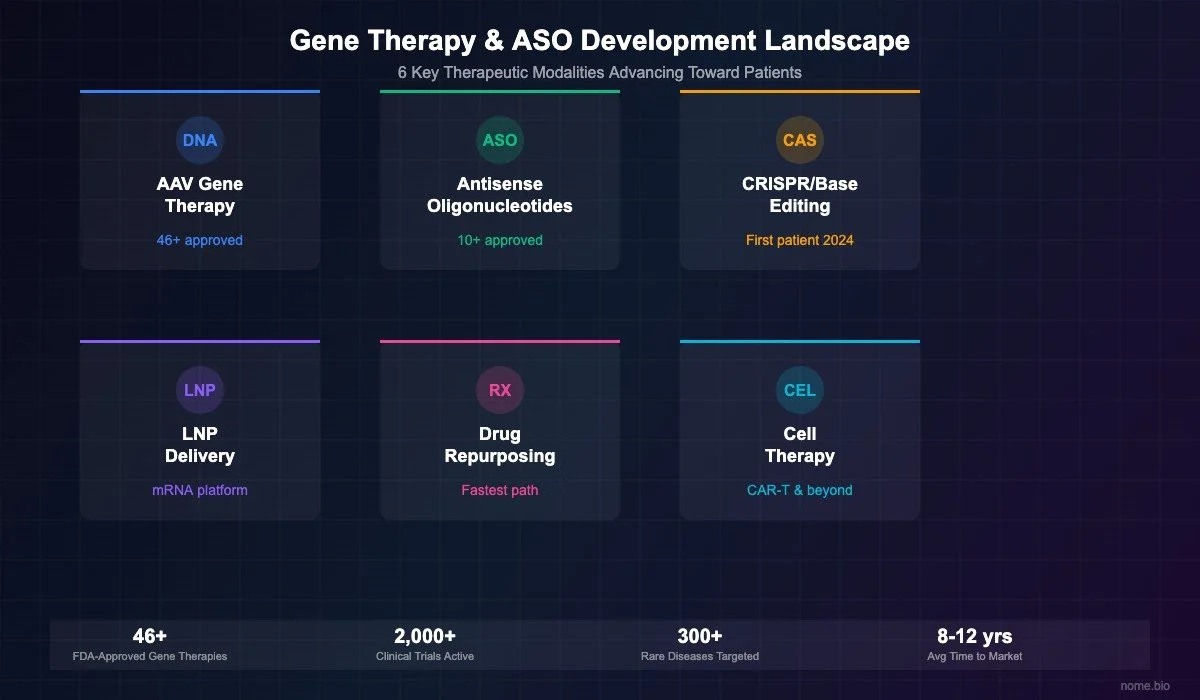
The FDA has now approved more than 46 gene and cell therapy products. That number has grown dramatically in the last few years. From 2017 to 2020, only a handful of gene therapies were approved each year. Starting in 2021, the pace picked up. In 2023 and 2024, the FDA approved 8 new gene and cell therapies each year, the highest rate in the history of the field.
On the ASO and RNA therapy side, 14 antisense oligonucleotide and siRNA drugs have received FDA approval. The most recent was olezarsen (Tryngolza), approved in December 2024 for familial chylomicronemia syndrome. Several more RNA therapies are expected to reach approval in 2025 and 2026, including donidalorsen for hereditary angioedema and plozasiran for severe hypertriglyceridemia.
Taken together, there are now more than 60 FDA-approved gene, cell, and RNA therapies available to patients. A decade ago, there were fewer than 10.
How Many Are in Clinical Trials Right Now?
Excluding early-stage lab research, there are more than 1,200 gene, cell, and RNA therapies currently in clinical trials with human patients, from Phase 1 through approval. The FDA alone has more than 2,500 active investigational new drug (IND) applications for cell and gene therapies, with approximately 1,300 specifically for gene therapies. An additional 35 programs are in Phase 3 pivotal trials, and 6 are in pre-registration awaiting FDA decisions.
What Diseases Are Being Targeted?
One of the most important trends for rare disease families: the pipeline is rapidly diversifying beyond cancer. Cancer (particularly CAR-T cell therapies for blood cancers) still makes up about 49% of clinical-stage gene therapies. But the balance is shifting fast.
Rare genetic and metabolic diseases now account for approximately 18% of clinical-stage programs, roughly 220 therapies in human trials. Hematology (blood disorders like sickle cell disease and hemophilia) accounts for another 10%. Neurological conditions, including rare neurodegenerative and neuromuscular diseases, make up 8%. Ophthalmology (inherited retinal diseases) is at 5%, and cardiovascular conditions at 4%.
The critical number: 51% of new gene therapy trials initiated in 2025 were for non-cancer conditions, up from 39% just one year earlier. This 12-point shift represents hundreds of new programs targeting the kinds of diseases that rare disease families live with. And 88% of all novel gene and cell therapies approved in 2024 had Orphan Drug designations, confirming that rare disease is the primary driver of approvals.
For families: if your child has a rare genetic disease affecting the blood, liver, brain, eyes, or metabolism, there are more clinical-stage programs targeting these conditions right now than at any point in history. The question is whether any of them match your child's specific gene and mutation.
Which Recent Approvals Matter Most for Rare Disease Families?
Not all approvals are equal. Some represent genuine breakthroughs that change what is possible for families. Here are the most significant from 2023 through early 2025.
Casgevy (December 2023) was the first CRISPR gene editing therapy ever approved by the FDA. It treats sickle cell disease and beta-thalassemia by editing blood stem cells outside the body. 93.5% of sickle cell patients in the clinical trial were free from severe pain crises for 12 or more months.
Waskyra (December 2025) is a milestone for a different reason: it was developed and brought to FDA approval by Fondazione Telethon, an Italian nonprofit. This is the first gene therapy from a non-profit sponsor to receive FDA approval. It treats Wiskott-Aldrich syndrome. Severe infections were reduced by 93%. This proves that foundations and nonprofits, not just pharma companies, can take therapies all the way to approval.
Baby KJ's personalized base editing (February 2025, investigational) demonstrated that a custom gene editing therapy could be designed, manufactured, and delivered to a single patient in six months. The CHOP/Penn/IGI team is now planning to expand to five more patients.
Beqvez (approved April 2024, discontinued February 2025) is the cautionary tale. Pfizer's AAV gene therapy for hemophilia B received FDA approval but zero patients ever received it commercially. Pfizer pulled it from the market, highlighting the challenge that even approved gene therapies face in adoption.
The Role of Foundations: Who Is Funding These Therapies?
One of the least understood aspects of rare disease drug development is how many programs are driven not by pharmaceutical companies, but by patient foundations, family-led nonprofits, and public-private partnerships. When a disease is too rare for pharma to justify the investment, foundations step in. And increasingly, they are succeeding.
Fondazione Telethon spent decades developing Waskyra at the San Raffaele Telethon Institute for Gene Therapy in Milan. When Orchard Therapeutics (a commercial company) abandoned the program during a 2022 restructuring, Telethon reacquired the rights and carried it to FDA approval. Their CEO, Ilaria Villa, described the model as one that "turns research into real treatments, especially where the market fails to act."
n-Lorem Foundation, founded by Stanley Crooke (the founder of Ionis Pharmaceuticals, the company behind most approved ASOs), develops free individualized ASO therapies for patients with nano-rare diseases. They have filed 13 or more INDs and have a pipeline covering more than 90 target genes. n-Lorem provides these therapies at no cost to patients or their insurance.
The Bespoke Gene Therapy Consortium (BGTC) is a $97 million public-private partnership between the NIH, the FDA, 12 pharmaceutical companies, and 10 nonprofit organizations. They have selected 8 rare diseases for standardized gene therapy clinical trials and are building shared manufacturing standards and regulatory templates that any developer can use.
CureSPG50 and Elpida Therapeutics, founded by Terry Pirovolakis, the father of a child with SPG50, developed a gene therapy that was selected for the BGTC clinical trial portfolio. This is one of a growing number of family-founded nonprofits that have taken programs into clinical-stage development.
The Cystic Fibrosis Foundation model remains the gold standard: CFF invested $150 million in Vertex Pharmaceuticals, which led to the development of Trikafta, now a $9 billion-per-year drug that treats 90% of CF patients. This venture philanthropy approach, where a foundation funds a company in exchange for a share of future revenue, has been replicated for dozens of other diseases.
For families: you do not need a pharmaceutical company to develop a therapy for your child's disease. The path through a foundation, a family-led nonprofit, or a public-private consortium is viable and has been proven to work all the way to FDA approval.
Why Are So Many Gene Therapies Getting Rejected?
In 2024 and 2025, several high-profile gene and cell therapies received Complete Response Letters (CRLs) from the FDA, meaning they were rejected in their current form. The reasons reveal an important pattern.
The science is rarely the problem. 74% of rejections from 2020 to 2024 were driven by manufacturing and quality issues. 56% cited facility inspection problems. 40% of early-stage applications (INDs) are being stopped due to manufacturing documentation deficiencies.
The biology works. Gene therapies and ASOs are doing what they are designed to do in clinical trials. What is breaking is the manufacturing infrastructure: GMP production, analytical methods, quality documentation, and process validation. These are operational challenges, not scientific ones.
For families, this has a practical implication: the team that manufactures and coordinates your child's therapy matters as much as the team that designs it. This is also why ASOs have an advantage for individualized programs. ASO manufacturing at small scale is simpler, well established, and validated through 14 approved products.
What to Watch in 2026
The most significant regulatory development: on February 23, 2026, the FDA published its "plausible mechanism" framework for individualized therapies. This applies to gene editing and ASO approaches and creates a formal pathway where data from as few as one patient can support full FDA approval, provided the scientific rationale, target engagement, and clinical improvement meet defined standards.
On the clinical side, the CHOP/Penn team is expanding personalized base editing to five more patients across seven urea cycle disorder genes. Prime Medicine is expected to start the first prime editing clinical trial. Nanoscope Therapeutics is pursuing the first gene-agnostic gene therapy for retinitis pigmentosa. And several foundation-backed programs in the BGTC portfolio are entering their first-in-human trials.
What This Means for Your Family
The field is growing and rare disease is leading. 51% of new gene therapy trials are for non-cancer diseases. 88% of approved therapies have orphan drug designations.
Foundations are proving the path works. From Fondazione Telethon to n-Lorem to CureSPG50, nonprofits are taking therapies from concept to patients, including to FDA approval.
ASOs are the most mature modality for individualized therapy. 14 approved products, well-established manufacturing, and a new FDA framework supporting individual patient programs.
Manufacturing is the bottleneck, not science. 74% of FDA rejections are manufacturing-related. Coordination and operational expertise matter as much as the biology.
The right answer depends on your child's specific mutation. No single modality is right for every disease. The only way to know which applies is to evaluate the mutation against every option in parallel.
What Nome Does
Nome tracks every approval, trial readout, CRL, and regulatory change, and evaluates how each one affects your child's options. Our free evaluation takes your child's genetic diagnosis and assesses it against every available modality: ASOs, gene therapy, gene editing, clinical trials, and drug repurposing. Within 1-2 weeks, you receive a clear report explaining which options are realistic, what the timeline looks like, and what the next steps are.
The field is moving fast. More families have more options today than at any point in history. The first step is always the same.
Submit your child's genetic diagnosis for a free evaluation at nome.bio.
REFERENCES
1. ASGCT Quarterly Industry Landscape Report, Q4 2025.
2. BioInformant. "List of U.S. FDA Approved Cell and Gene Therapy Products." November 2025.
3. FDA. "Approved Cellular and Gene Therapy Products." fda.gov.
4. The FDA Group. "Behind the Rejections: Analysis of 89 FDA CRLs." September 2025.
5. Drug Discovery News. "Why gene and cell therapies are stalling at the FDA." August 2025.
6. Fondazione Telethon. "FDA Approval of Waskyra." Press release, December 10, 2025.
7. FNIH. "BGTC Clinical Trial Portfolio." fnih.org, May 2023.
8. CGTLive. "Top FDA Gene and Cell Therapy News: 2025 Year-End Recap." December 2025.
9. FDA. "Framework for Individualized Antisense Oligonucleotide Drug Products." February 23, 2026.
10. Jonker AH, et al. Nature Reviews Drug Discovery. 2025;24:40-56.
This article is for informational purposes only and does not constitute medical or investment advice.